
Crosslinked
UHMWPE
for THR
It is widely recognized that early loosening of prosthetic components attributable to polyethylene wear debris is one of the main causes of failure in the medium-to-long term. In the prosthetic components, due to UHMWPE deformability, wear debris are of micrometric dimensions elongated in shape (fibrils) and of nanometric dimensions, more spheroid in shape (nanoparticles). Recently, crosslinking technologies have been developed, recognising four different steps:
Radiation with gamma or beta rays at dose levels between 50 and 100 kGy, in order to achieve crosslinking.
Heating at temperatures between 80°C and 160°C, to eliminate the residual free radicals generated during radiation.
Machining of the component, which results in the removal of the superficial oxidised layer.
Final sterilisation without radiation to avoid further alteration to the structure.
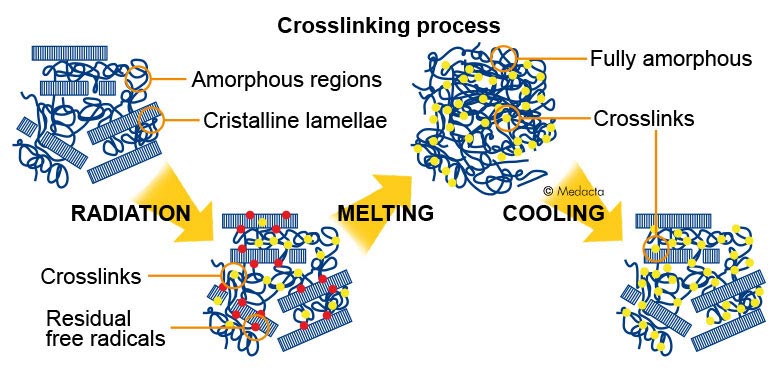
All UHMWPE types processed as above, have the following features (though with some differences related to treatment conditions):
extremely low wear rates;
absence of aging related phenomena and are indeed of great interest for clinical use.
Clinical trials are ongoing and the first positive preliminary results are now available.[1]
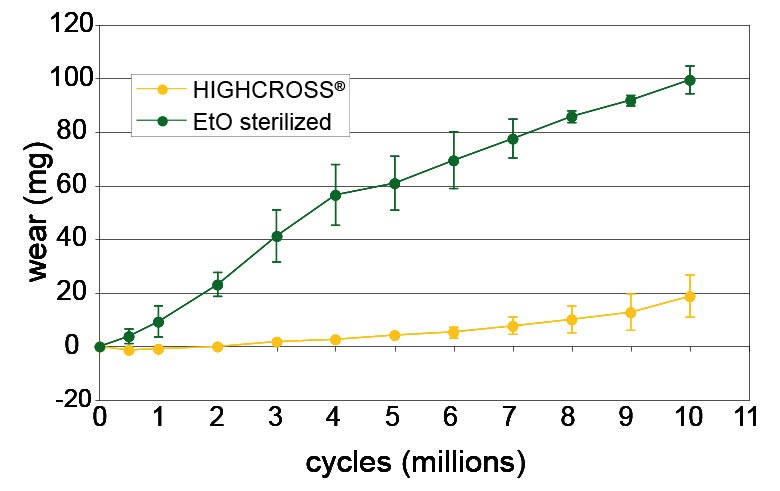
After 10 million wear cycles on a hip joint simulator, the crosslinked UHMWPE wear rate is nearly zero and there is a significant difference between the 25 kGy sterilised and the crosslinked material wear rate. Therefore, after artificial aging, there is no difference in the wear rate for crosslinked UHMWPE.[5]
Wear test results from a hip joint simulator after 10 million cycles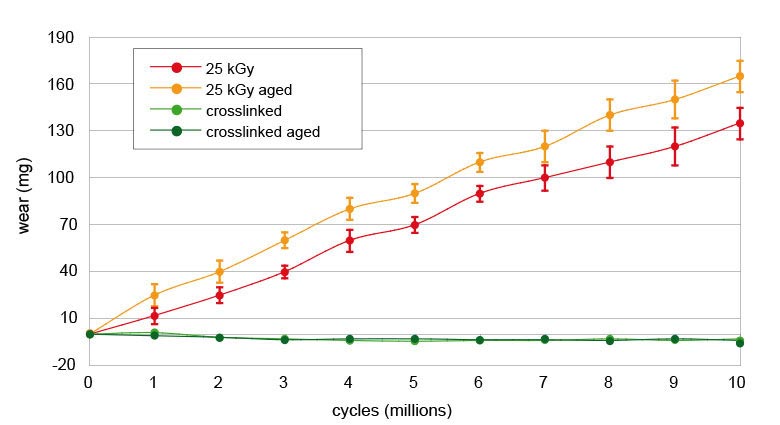
Different types of crosslinked UHMWPE are commercially available and are characterised by:
Not all crosslinked UHMWPE provides adequate performance, mainly due to low radiation levels, low stabilisation temperatures and final sterilisation with gamma rays. HIGHCROSS by Medacta is characterised by:
Such innovative production strategies yield a high quality material with enhanced properties.
Medacta has started long term clinical studies to confirm the excellent in vitro and first short-term results.
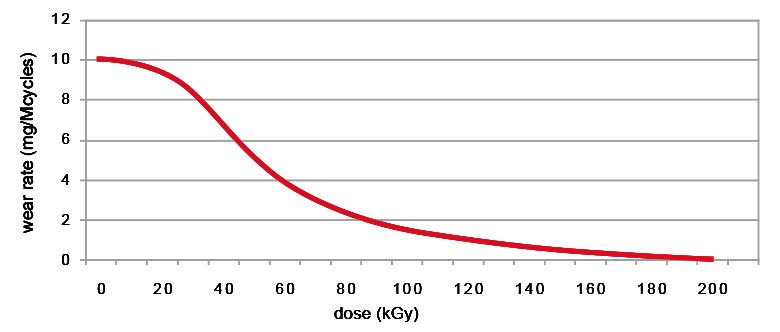
It has been proven that wear resistance increases proportionally to the radiation level, reaching very low wear values after 100 kGy exposure.
50 kGy radiation eliminates the micrometric wear particles, but not the nanometric particles which remain abundant.
Only with radiation levels of at least 100 kGy do the number of nanoparticles decrease when compared to the quantity produced by traditional UHMWPE[2].
Pin on disk tests: effect or radiation dose[2]
Hip simulator tests: effect of crosslinking conditions[2]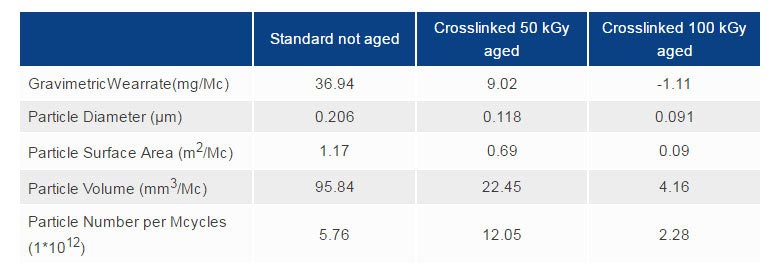
Crosslinking occurs during irradiation (in air or nitrogen) but there is also concomitant formation of free radicals.
25 kGy gamma rays sterilisation results in oxidation both before and after implantation (even if sterilisation occurs in nitrogen). Oxidation causes weakening of the material with a drastic decrease in its mechanical properties.
If after radiation the material is stabilised with heat treatment, free radicals can be eliminated.
Complete elimination of free radicals is achieved only if the temperature rises over 135°C (the melting point of UHMWPE)[3].
In this way, the mechanical properties of cross linked and stabilised UHMWPE remain unaltered over time and are, therefore, superior to those of standard UHMWPE, which deteriorate over times.[3]
High crosslinking improves the wear resistance of UHMWPE, but causes a slight decrease in other mechanical properties.
For the same radiation level, these mechanical properties are strongly influenced by the crystallinity of the UHMWPE, which can vary between 42% and 54%. Only careful control of the manufacturing process, particularly of the cooling rate, provides the best compromise for crosslinked UHMWPE between wear resistance and other mechanical properties.[3]
After the crosslinking process and machining, the final implant has to be sterilised.
It is evident, that in order to prevent new free radical formation and subsequent oxidation, an inert final sterilisation process with ethylene oxide (or gas plasma) must be used.
Any sterilisation process involving gamma rays (even in an inert environment, such as nitrogen or vacuum) must be avoided[4].
Regarding wear resistance, it has been widely demonstrated that crosslinked UHMWPE wear is not measurable using normal test parameters. For this reason, Medacta performed wear tests, simulating critical conditions, matching HIGHCROSS with CoCrMo heads abraded with hydroxyapatite powder to increase their roughness. The results show that HIGHCROSS treated liners have significantly greater wear resistance than standard UHMWPE liners.
Optical microphotographs of the worn surface after 10 million cycles show that standard UHMWPE liners have no machining marks left (due to the wear process). On the contrary HIGHCROSS liners still show the lathe machining marks.[5]


Regarding the mechanical properties, after artificial aging, HIGHCROSS better maintains its mechanical properties compared to a 25 kGy gamma ray sterilised UHMWPE.
[1] Currier, B.H:, Wittman, M.W., “Retrieval Analysis of Crosslinked Acetabular Bearings”, Crosslinked and Thermally treated UHMWPE for Joint Replacements STP1445, ASTM 2004
[2] Scott, M., Widding, K., Ries, M., Shanbhag, A., Smith & Nephew, Inc. Memphis. "Wear Particle Analyses of Conventional and Crosslinked Ultra High Molecular Weight Polyethylene Tested in an Anatomic Hip Simulator" 47th Annual Meeting, ORS, Feb. 25-28, 2001, San Francisco, California
[3] Chiesa R, Moscatelli M, Giordano C, Siccardi F, Cigada A, 2004 “Influence of heat treatment on structural, mechanical and wear properties of crosslinked UHMWPE”. Journal of Applied Biomaterials & Biomechanics 2, 20-28.
[4] Muratoglu, O.K., Bragdon, C.R., O’Connor, D.O., Delaney, J., Jasty, M., Harris, W.H., 2000. “The comparison of the wear behaviour of four types of crosslinked acetabular components”, 46th Annual Meeting, ORS, March 12-15 2000, Orlando, Florida.
[5] Data on files Medacta